Bladder cancer has moved up from the 10th to the 9th most diagnosed cancer worldwide, with both incidence and mortality rates increasing. The new data estimated that 614,298 people were diagnosed with bladder cancer in 2022 around the world, marking a 7.1% increase from the data reported in 2020. The new 5-year prevalence also shows that 1,950,315 people (all genders) are living with bladder cancer within five years of a past diagnosis. Focusing on gender-specific data, it estimates 523,674 new bladder cancer cases in men, representing 5.4% of all new cancer cases in men globally, making it the 6th most common cancer among men. [1]
In Saudi Arabia, among 1,791 genitourinary (GU) tumor cases were identified. Bladder cancer was the most prevalent (42%), followed by prostate cancer (21%) and renal cell carcinoma (21%). [2]. 82.7% of bladder cancer cases are found to be in men, while 17.3% are in women. Transitional cell carcinoma was the most common histological type, followed by adenocarcinoma and squamous cell carcinoma. Of the transitional cell carcinoma cases, the most common type is superficial, followed by muscle-invasive. [3] In a comprehensive analysis from the Najran region, a significant male predominance, with a high rate of advanced-stage presentation, and substantial survival disparities were observed. [4]
The most common types of bladder cancer are urothelial bladder cancer, comprising 90% of bladder cancer, and other non-urothelial bladder cancer, such as Squamous cell bladder cancer, Adenocarcinoma bladder cancer and other rare cancer. [5]
Smoking is the most important risk factor for bladder cancer, increasing the incidence and mortality-related bladder cancer. [6] Not only traditional cigarette smoking but also electronic cigarettes have been linked to the increased incidence and progression of bladder cancer. [7] Since tobacco contains many known carcinogenic compounds, including aromatic amines and N-nitroso compounds, which are excreted in the urine and therefore come into close contact with the bladder lining, resulting in DNA adducts that can induce different mutations [8].
High fluid intake reduces the risk of bladder cancer by diluting the urinary concentrations of lifestyle- or environment-related metabolites or by decreasing the duration of exposure of the bladder epithelium to such compounds owing to more frequent voiding. Conversely, high fluid intake might increase the risk if the fluids contain carcinogens such as arsenic, simply because of a higher cumulative dose of carcinogens [9].
On the other hand, green tea and its polyphenols may lower the risk of bladder cancer due to their strong antioxidant and anticarcinogenic effects. [10, 11] Additionally, fruits and vegetables may reduce bladder cancer risk due to their antioxidant properties. [12] Following the Mediterranean diet, which features a high intake of fruits, vegetables, legumes, and cereals; moderate to high consumption of fish; low to moderate intake of milk and dairy products; and limited intake of meat and meat products, reduces the risk of developing bladder cancer [13].
Diabetes and obesity negatively influence bladder cancer prognosis and outcomes, in addition to increased risk of mortality. [14]
Six categories of carcinogenic exposures in the workplace have been reported in the IARC monographs as risk factors for bladder cancer: ortho-toluidine; arsenic and inorganic arsenic compounds; X-ray radiation and γ-radiation; 2-naphthylamine; 4-aminobiphenyl; and benzidine [15]. Exposures to benzene in the petroleum stream, toluene, and xylene among shoe machine operators, tenders, automobile mechanics, and other textile machine operators may be linked to an increased risk of bladder cancer [16, 17].
Numerous genetic factors significantly influence the risk of developing bladder cancer. Notably, mutations in several key chromosomal genes, including FGFR3, RB1, HRAS, TP53, and TSC1, can elevate this risk. Alterations in the genes located on human chromosome 9 are often linked to tumor formation in bladder cancer, while mutations on chromosome 22 may also contribute to the disease. Research indicates that approximately 70% of bladder cancers harbor a specific mutation in the telomerase reverse transcriptase (TERT) gene, underscoring its role in bladder cancer. Additionally, HRAS, being a proto-oncogene, has the potential to lead to cancer development in various organs, including the bladder. [18]
Chronic bladder irritation includes chronic or recurrent urinary tract infection, chronic indwelling urinary catheter, bladder calculi, foreign bodies, intravesical Bacillus Calmette-Guerin (BCG), and prolonged exposure to cyclophosphamide, all of which are associated with increased bladder cancer risk. [19] Human papillomavirus (HPV) may play a role in bladder carcinogenesis and contribute to a worse prognosis for patients with bladder cancer, especially men [20].
Participants with a first-degree relative who has bladder cancer had nearly twice the risk of developing the disease. The risk was also higher when participants reported a first-degree relative with a history of female genital cancer, melanoma, or tobacco-related cancers. [21]
Squamous cell carcinoma (SCC) and adenocarcinoma of the bladder typically present with nonspecific lower urinary tract symptoms that overlap with other bladder cancer subtypes. Current evidence does not support symptom-based differentiation between these histologies. Definitive diagnosis relies on cystoscopic evaluation and histopathological confirmation, rather than clinical presentation alone.
Regardless of tumor histology (urothelial carcinoma, SCC, or adenocarcinoma), patients most frequently present with:
Hematuria (microscopic or gross), the most common presenting symptom [22, 23].
Irritative voiding symptoms, including urinary frequency, urgency, dysuria, nocturia, and burning on micturition [22, 23].
Pelvic, suprapubic, or lower abdominal pain [22, 24].
Obstructive urinary symptoms, such as weak urinary stream, difficulty voiding, urinary retention, and, in advanced cases, hydronephrosis [22, 23].
Systemic manifestations of advanced disease, including weight loss, bone pain, abdominal pain, or symptoms related to metastatic spread [25].
While not diagnostic, SCC of the bladder is more frequently associated with:
A background of chronic bladder irritation, including long-term catheterization, spinal cord injury, bladder stones, recurrent urinary tract infections, or schistosomiasis [22, 23, 26].
High rates of hematuria, with a substantial proportion of patients also reporting irritative urinary symptoms [22-24].
Concomitant chronic cystitis or active urinary tract infection at the time of diagnosis [23, 26].
Similarly nonspecific, adenocarcinoma of the bladder is often described with:
Hematuria accompanied by irritative symptoms, such as urinary frequency, dysuria, and suprapubic discomfort [27, 28].
In select cases—particularly mucinous variants or tumors involving the bladder neck or outlet—voiding difficulty or bladder outlet obstruction, which may occur with minimal or absent hematuria [27].
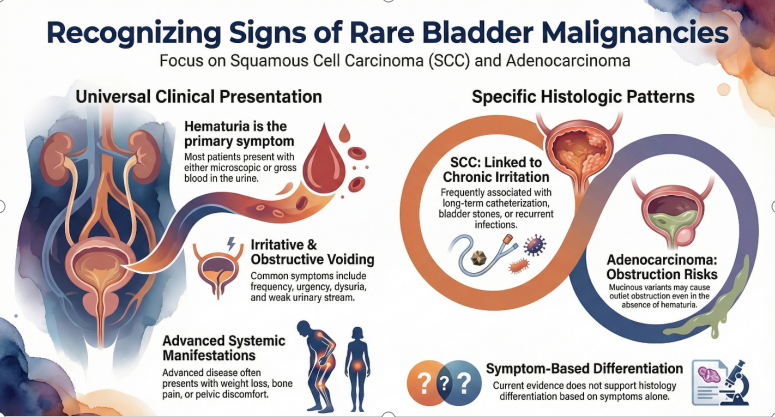
Figure 1: Clinical presentation of Squamous cell carcinoma (SCC) and adenocarcinoma of the bladder.
Squamous cell carcinoma (SCC) and adenocarcinoma of the bladder are both diagnosed through histopathological evaluation of tissue specimens obtained via cystoscopy or surgical resection. No imaging modalities or urine-based tests can independently distinguish these histologic subtypes. Differentiation relies on morphologic assessment, supported by clinical context and selected immunohistochemical (IHC) markers.
The initial diagnostic approach is similar for both entities and includes:
Cystoscopy with biopsy or transurethral resection of bladder tumor (TURBT) following evaluation of hematuria; histology remains the diagnostic gold standard for all bladder cancers [26].
Cross-sectional imaging (CT or MRI)—including structured systems such as VI-RADS—used for staging and assessment of local or distant spread rather than histologic classification [29, 30].
Additional evaluation in suspected adenocarcinoma, aimed at excluding secondary involvement from colorectal, prostatic, gynecologic, or urachal primaries through targeted imaging and clinical work-up [28, 31].
Definitive differentiation is based on characteristic microscopic findings:
Squamous cell carcinoma is defined by a pure squamous phenotype, including keratin pearl formation, intercellular bridges, and keratin debris, with no urothelial component [23, 26, 31].
Adenocarcinoma is characterized by true glandular differentiation, often with mucin production, and may exhibit enteric, mucinous, signet-ring, or mixed growth patterns [28, 31, 32].
Tumors containing any urothelial component are classified as urothelial carcinoma with squamous or glandular differentiation, rather than pure SCC or adenocarcinoma [28, 31].
The diagnosis of primary bladder adenocarcinoma is reserved for tumors with pure non-urothelial glandular morphology [28, 31].
Immunohistochemistry serves as a supportive tool, not a standalone diagnostic method:
In SCC, markers such as p40, p63, and CK5/6 confirm squamous differentiation but do not reliably distinguish pure SCC from urothelial carcinoma with squamous differentiation [23, 28, 31].
In adenocarcinoma, marker panels (e.g., CK7, CK20, CDX2, SATB2, β-catenin) assist in differentiating primary bladder adenocarcinoma from metastatic colorectal or Müllerian tumors [31-33].
Molecular and genomic profiling has demonstrated distinct mutational and immune-oncology signatures across bladder cancer subtypes; however, these approaches are not routinely required for basic histologic classification in current practice [32, 34].
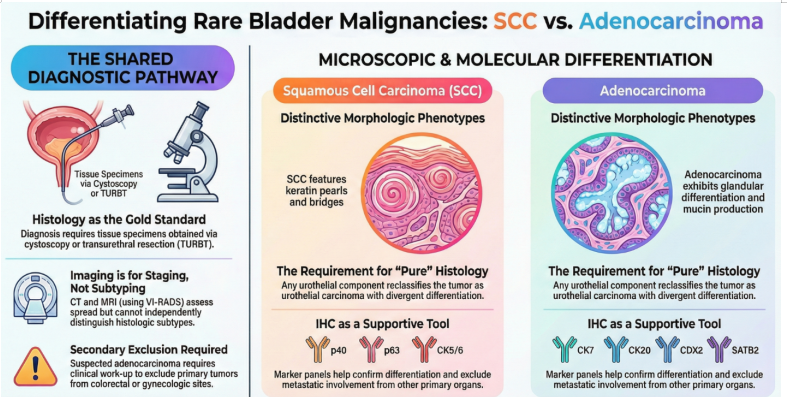
Figure 2: Diagnostic differentiation between SCC and adenocarcinoma of the bladder.
Current treatments for bladder SCC mainly involve surgery, as systemic options are limited. For localized, non-metastatic disease, radical cystectomy with pelvic lymph node dissection is the standard, providing better survival than radiotherapy or chemotherapy. Radiotherapy, alone or combined, is reserved for unfit patients but yields worse outcomes. The carcinoma often resists chemotherapy, with no clear survival benefit from neoadjuvant or adjuvant therapies over surgery. In advanced cases, prognosis is poor, and treatment is usually palliative, often following urothelial carcinoma protocols. Emerging data suggest some activity of immune checkpoint inhibitors, especially before surgery, but evidence is limited. Early surgery and participation in clinical trials are strongly advised.
Radical cystectomy with pelvic lymph node dissection is widely regarded as the reference standard for the management of localized SCC and is associated with the most favorable survival outcomes compared with non-surgical approaches [36]. Evidence from large population-based analyses, systematic reviews, and institutional series consistently demonstrates that radical cystectomy, performed with or without perioperative chemotherapy, confers superior overall and cancer-specific survival compared with chemotherapy alone, radiotherapy alone, or combined chemoradiation in patients with nonmetastatic disease [37]. Long-term outcomes following surgery highlight the prognostic importance of tumor stage and nodal involvement, with durable disease-free survival reported in appropriately selected patients [37-39].
The benefit of peri-operative chemotherapy in node-negative localized SCC remains unproven, and available data suggest limited chemosensitivity in pure squamous histology, supporting a surgery-first approach in resectable disease [40]. Bladder-preserving strategies, including chemoradiation, have been explored in small retrospective series but are generally reserved for patients who are medically unfit for radical surgery or who decline cystectomy, given the weaker and less consistent supporting evidence [41]. Partial cystectomy is considered only in highly selected cases with favorable anatomy and requires stringent long-term surveillance [41]. Overall, current evidence supports early radical cystectomy with lymphadenectomy as the guideline-concordant primary treatment for non-metastatic bladder SCC, with non-surgical alternatives limited to selected clinical scenarios [42].
For patients with localized or locally advanced SCC of the bladder, surgery, particularly radical cystectomy with pelvic lymph node dissection, represents the cornerstone of curative treatment, while other therapeutic modalities generally serve adjunctive or alternative roles [37, 43]. Large population-based analyses from SEER and the National Cancer Database consistently demonstrate that radical surgery is associated with superior overall and cancer-specific survival compared with radiotherapy alone, observation, or other non-surgical approaches in non-metastatic disease [44]. Survival benefits with surgery are observed in both localized and regional (locally advanced) stages, although the magnitude of benefit decreases in the presence of distant metastases, where surgery is largely palliative [44, 45].
Bladder-preserving approaches, including partial cystectomy or TURBT-based strategies, have been reported only in highly selected cases and are supported primarily by limited retrospective data [46]. Long-term outcomes from population studies indicate that these approaches generally provide inferior cancer-specific survival compared with radical cystectomy and require intensive lifelong surveillance [36].
The addition of perioperative systemic therapy has not shown a clear survival benefit in node-negative localized SCC and is therefore not routinely recommended; however, in patients with very high-risk features—such as node-positive, T4b, or metastatic disease—combined surgical and systemic approaches may reduce cancer-specific mortality [37, 40, 47]. In bilharzial-associated SCC, selected series suggest a potential role for perioperative radiotherapy to improve local control, though the quality of evidence remains limited. Overall, early radical cystectomy remains the most effective curative strategy for localized and locally advanced bladder SCC, with non-surgical options reserved for carefully selected or surgically unfit patients [26, 36].
For bladder SCC, radical cystectomy should almost always include bilateral PLND, as omission clearly worsens survival. Pelvic lymph node dissection (PLND) clearly improves survival in bladder SCC when performed with cystectomy, but current data support a “standard” rather than an extended template. In a SEER analysis of variant histology, PLND versus no PLND significantly improved overall survival in SCC (HR ~0.65), with median OS nearly doubled (≈71 vs 37 months) [48]. A large NCDB study of variant bladder cancers confirmed that lymph node dissection is independently associated with better OS in the squamous subtype (HR 0.50), one of the strongest benefits among variants [49]. Despite this, SCC has high pNx rates (~68%), indicating PLND is underused in this histology [49].
Evidence on extent comes from mixed histology (mostly urothelial) MIBC, but no data suggest SCC behaves differently:
Two RCTs and multiple meta-analyses show no improvement in overall or disease-free survival with extended PLND to the aortic bifurcation compared with standard PLND up to the common iliac bifurcation [50, 51].
Extended PLND increases major complications, lymphoceles, and 90-day mortality [50].
SEER analysis in variant histology found greater PLND extent did not reduce cancer-specific mortality in VHBC (including SCC), although it improved staging yield [52].
For variant histologies, PLND remains prognostic even when chemo or neoadjuvant therapy is used, but added survival from “more extensive” dissection diminishes after neoadjuvant chemotherapy [53]. Immunotherapy outcomes (pembrolizumab) do not appear improved by more extensive node harvest, and >14 nodes after NAC may even correlate with worse progression-free survival in mixed histology cohorts [54].
Definitive radiotherapy is feasible for non‑surgical bladder SCC, but expected cure rates are low; concurrent chemoradiation is preferable to RT alone when possible. Non-bilharzial SCC of the bladder is aggressive and usually treated with radical cystectomy. For patients who cannot undergo surgery, evidence for definitive radiotherapy (RT) is limited and mostly retrospective, but offers a potentially curative alternative to best supportive care.
In the largest NCDB analysis of localized cT2–3N0 pure SCC, RT alone and chemoradiation (CRT) had substantially worse overall survival (OS) than cystectomy (HR for death vs RC: RT 4.78; CRT 1.61) [37]. A SEER study of 5653 SCC cases found median survival 12 months with RT, 9 months with chemotherapy, vs markedly better with surgery; adding RT to surgery did not improve OS, likely reflecting selection bias [44]. Historical RT alone series in SCC report ~16% 3 year disease free survival with 60–65 Gy, indicating poor local control [55].
NCDB CRT analysis (79 SCC, cT2–T4N0M0) using concurrent Chemoradiation (CRT) found median OS ~15 months for SCC vs ~29–30 months for urothelial carcinoma; SCC histology independently predicted worse OS [55]. Case-based and small series suggest some long-term survivors using 5 FU/mitomycin or cisplatin-based CRT, extrapolating from anal SCC and urothelial CRT trials [36, 55].
Guidelines for MIBC support definitive RT or CRT in cystectomy-unfit patients, acknowledging lower-level evidence, and this is often extrapolated to SCC [56]. Narrative and systematic reviews emphasize that RT with concomitant chemotherapy is a reasonable alternative for unresectable tumors or in patients seeking bladder preservation, but data specific to SCC are sparse and of low level [36]. In broader MIBC cohorts unfit for surgery or cisplatin, RT (often without chemo) substantially improves OS vs supportive care, supporting its use as active treatment in frail patients [57].
In bladder SCC patients not suitable for cystectomy, a bladder-preserving approach with TURBT followed by chemoradiotherapy is preferred. Curative radiotherapy often involves doses ≥60 Gy with radiosensitizers like 5-fluorouracil, mitomycin C, cisplatin, or weekly gemcitabine. While this may control disease in some, cure rates are lower than with cystectomy [36]. For those unable to have chemotherapy, definitive radiotherapy alone at radical doses may improve survival versus no treatment, but outcomes are limited and palliative [44]. Due to high local recurrence risk, close surveillance with cystoscopy and imaging is vital, and salvage cystectomy should be considered if the patient’s condition and fitness improve [36, 55].
Adjuvant radiotherapy (ART) after radical cystectomy is being revisited because loco-regional recurrence rates for ≥pT3 disease remain high, and salvage is difficult. Modern intensity modulated techniques have reduced historical toxicity, enabling safer postoperative treatment. High risk features associated with loco regional failure include pT3–4, pN+, positive margins, and limited lymph node dissection (<10 nodes) [58, 59]. Phase II–III and retrospective studies consistently show substantial reductions in pelvic relapse with ART or chemoradiotherapy compared with observation or chemotherapy alone, especially in ≥pT3/4 or margin-positive disease [60, 61].
With IMRT/VMAT, severe GI toxicity is markedly lower than in historical 2D RT: grade ≥3 GI events ~3–6%, though grade 2 GI symptoms are common. Phase II trials show acceptable urinary toxicity even in neobladder patients [62].
Reviews and guidelines suggest that ART is reasonable for selected high-risk MIBC, especially with positive margins, pT3–4, pN+, or suboptimal node dissection, and where access to salvage or immunotherapy is limited [63]. Large databases show no consistent OS benefit overall, but a possible benefit in marginally positive patients [64, 65]. International consensus CTV guidelines now recommend including the cystectomy bed and common iliac nodes for high-risk cases [66].
Most work on neoadjuvant radiotherapy (RT) in bladder cancer is in mixed or predominantly urothelial histology. For pure bladder squamous cell carcinoma (SCC), evidence is largely indirect, coming from older series or reviews that pool histologies. A systematic review of bladder SCC notes that preoperative RT + radical cystectomy (RC) appears to improve survival compared with RC alone, but the data are from small, older series and are insufficient for firm recommendations. The same review highlights SCC’s high local recurrence risk and suggests neoadjuvant or adjuvant RT (± systemic therapy) may reduce recurrence, but emphasizes the lack of randomized SCC-only trials [36].
Contemporary reviews of perioperative RT in muscle-invasive bladder cancer argue that preoperative RT is attractive in very high-risk, locally advanced disease to sterilize microscopic extension and improve pathologic response, but call the evidence “preliminary” and non-definitive [67]. Current neoadjuvant RT trials (often with immunotherapy such as nivolumab or durvalumab) are in urothelial carcinoma, not SCC; they explore feasibility and pathological complete response but do not yet guide SCC practice [68].
For operable, locally advanced SCC, RC remains standard; neoadjuvant RT can be considered only in selected very high-risk cases, extrapolating from mixed histology data and acknowledging the lack of SCC-specific proof [36, 38]. For patients unfit for RC, management usually shifts to definitive chemoradiation, not “neoadjuvant” RT, again extrapolated from urothelial MIBC paradigms rather than SCC trials [69].
For bladder squamous cell carcinoma, radical cystectomy remains standard; chemoradiation is mainly a bladder‑sparing or non‑surgical option and appears less effective for survival but can offer local control when surgery is not feasible.
A large NCDB study of 828 patients with localized muscle-invasive pure SCC (cT2–3N0M0) compared RC ± perioperative chemotherapy, chemotherapy alone, radiation alone, and definitive chemoradiation. RC (with or without chemo) had the best survival; chemoradiation carried a higher death risk (HR 1.61 vs RC alone) but was better than radiation alone or chemotherapy alone [37]. A systematic review of non-bilharzial SCC concluded RC is the gold standard; radiotherapy alone gives poor outcomes, while multimodality approaches (pre op RT + RC, or RC + systemic therapy) may help, but evidence is low-level and heterogeneous [36].
Variant histology (including SCC) shows worse survival after bladder preserving RT than pure urothelial carcinoma, highlighting more aggressive biology and the need for intensification or surgery when possible [55].
Indications of chemoradiation in Bladder SCC:
Regimens generally mirror urothelial protocols: conventional or hypofractionated pelvic RT (≈55–64 Gy) with concurrent cisplatin, gemcitabine, or 5 FU/mitomycin C as radiosensitizer [56]. Given chemotherapy resistance concerns in SCC, systemic control may still be suboptimal, and close surveillance or consideration of combined RC, where feasible, is important [36, 55].
Bladder cancer has moved up from the 10th to the 9th most diagnosed cancer worldwide, with both incidence and mortality rates increasing. The new data estimated that 614,298 people were diagnosed with bladder cancer in 2022 around the world, marking a 7.1% increase from the data reported in 2020. The new 5-year prevalence also shows that 1,950,315 people (all genders) are living with bladder cancer within five years of a past diagnosis. Focusing on gender-specific data, it estimates 523,674 new bladder cancer cases in men, representing 5.4% of all new cancer cases in men globally, making it the 6th most common cancer among men. [1]
In Saudi Arabia, among 1,791 genitourinary (GU) tumor cases were identified. Bladder cancer was the most prevalent (42%), followed by prostate cancer (21%) and renal cell carcinoma (21%). [2]. 82.7% of bladder cancer cases are found to be in men, while 17.3% are in women. Transitional cell carcinoma was the most common histological type, followed by adenocarcinoma and squamous cell carcinoma. Of the transitional cell carcinoma cases, the most common type is superficial, followed by muscle-invasive. [3] In a comprehensive analysis from the Najran region, a significant male predominance, with a high rate of advanced-stage presentation, and substantial survival disparities were observed. [4]
The most common types of bladder cancer are urothelial bladder cancer, comprising 90% of bladder cancer, and other non-urothelial bladder cancer, such as Squamous cell bladder cancer, Adenocarcinoma bladder cancer and other rare cancer. [5]
Smoking is the most important risk factor for bladder cancer, increasing the incidence and mortality-related bladder cancer. [6] Not only traditional cigarette smoking but also electronic cigarettes have been linked to the increased incidence and progression of bladder cancer. [7] Since tobacco contains many known carcinogenic compounds, including aromatic amines and N-nitroso compounds, which are excreted in the urine and therefore come into close contact with the bladder lining, resulting in DNA adducts that can induce different mutations [8].
High fluid intake reduces the risk of bladder cancer by diluting the urinary concentrations of lifestyle- or environment-related metabolites or by decreasing the duration of exposure of the bladder epithelium to such compounds owing to more frequent voiding. Conversely, high fluid intake might increase the risk if the fluids contain carcinogens such as arsenic, simply because of a higher cumulative dose of carcinogens [9].
On the other hand, green tea and its polyphenols may lower the risk of bladder cancer due to their strong antioxidant and anticarcinogenic effects. [10, 11] Additionally, fruits and vegetables may reduce bladder cancer risk due to their antioxidant properties. [12] Following the Mediterranean diet, which features a high intake of fruits, vegetables, legumes, and cereals; moderate to high consumption of fish; low to moderate intake of milk and dairy products; and limited intake of meat and meat products, reduces the risk of developing bladder cancer [13].
Diabetes and obesity negatively influence bladder cancer prognosis and outcomes, in addition to increased risk of mortality. [14]
Six categories of carcinogenic exposures in the workplace have been reported in the IARC monographs as risk factors for bladder cancer: ortho-toluidine; arsenic and inorganic arsenic compounds; X-ray radiation and γ-radiation; 2-naphthylamine; 4-aminobiphenyl; and benzidine [15]. Exposures to benzene in the petroleum stream, toluene, and xylene among shoe machine operators, tenders, automobile mechanics, and other textile machine operators may be linked to an increased risk of bladder cancer [16, 17].
Numerous genetic factors significantly influence the risk of developing bladder cancer. Notably, mutations in several key chromosomal genes, including FGFR3, RB1, HRAS, TP53, and TSC1, can elevate this risk. Alterations in the genes located on human chromosome 9 are often linked to tumor formation in bladder cancer, while mutations on chromosome 22 may also contribute to the disease. Research indicates that approximately 70% of bladder cancers harbor a specific mutation in the telomerase reverse transcriptase (TERT) gene, underscoring its role in bladder cancer. Additionally, HRAS, being a proto-oncogene, has the potential to lead to cancer development in various organs, including the bladder. [18]
Chronic bladder irritation includes chronic or recurrent urinary tract infection, chronic indwelling urinary catheter, bladder calculi, foreign bodies, intravesical Bacillus Calmette-Guerin (BCG), and prolonged exposure to cyclophosphamide, all of which are associated with increased bladder cancer risk. [19] Human papillomavirus (HPV) may play a role in bladder carcinogenesis and contribute to a worse prognosis for patients with bladder cancer, especially men [20].
Participants with a first-degree relative who has bladder cancer had nearly twice the risk of developing the disease. The risk was also higher when participants reported a first-degree relative with a history of female genital cancer, melanoma, or tobacco-related cancers. [21]
Squamous cell carcinoma (SCC) and adenocarcinoma of the bladder typically present with nonspecific lower urinary tract symptoms that overlap with other bladder cancer subtypes. Current evidence does not support symptom-based differentiation between these histologies. Definitive diagnosis relies on cystoscopic evaluation and histopathological confirmation, rather than clinical presentation alone.
Regardless of tumor histology (urothelial carcinoma, SCC, or adenocarcinoma), patients most frequently present with:
Hematuria (microscopic or gross), the most common presenting symptom [22, 23].
Irritative voiding symptoms, including urinary frequency, urgency, dysuria, nocturia, and burning on micturition [22, 23].
Pelvic, suprapubic, or lower abdominal pain [22, 24].
Obstructive urinary symptoms, such as weak urinary stream, difficulty voiding, urinary retention, and, in advanced cases, hydronephrosis [22, 23].
Systemic manifestations of advanced disease, including weight loss, bone pain, abdominal pain, or symptoms related to metastatic spread [25].
While not diagnostic, SCC of the bladder is more frequently associated with:
A background of chronic bladder irritation, including long-term catheterization, spinal cord injury, bladder stones, recurrent urinary tract infections, or schistosomiasis [22, 23, 26].
High rates of hematuria, with a substantial proportion of patients also reporting irritative urinary symptoms [22-24].
Concomitant chronic cystitis or active urinary tract infection at the time of diagnosis [23, 26].
Similarly nonspecific, adenocarcinoma of the bladder is often described with:
Hematuria accompanied by irritative symptoms, such as urinary frequency, dysuria, and suprapubic discomfort [27, 28].
In select cases—particularly mucinous variants or tumors involving the bladder neck or outlet—voiding difficulty or bladder outlet obstruction, which may occur with minimal or absent hematuria [27].

Figure 1: Clinical presentation of Squamous cell carcinoma (SCC) and adenocarcinoma of the bladder.
Squamous cell carcinoma (SCC) and adenocarcinoma of the bladder are both diagnosed through histopathological evaluation of tissue specimens obtained via cystoscopy or surgical resection. No imaging modalities or urine-based tests can independently distinguish these histologic subtypes. Differentiation relies on morphologic assessment, supported by clinical context and selected immunohistochemical (IHC) markers.
The initial diagnostic approach is similar for both entities and includes:
Cystoscopy with biopsy or transurethral resection of bladder tumor (TURBT) following evaluation of hematuria; histology remains the diagnostic gold standard for all bladder cancers [26].
Cross-sectional imaging (CT or MRI)—including structured systems such as VI-RADS—used for staging and assessment of local or distant spread rather than histologic classification [29, 30].
Additional evaluation in suspected adenocarcinoma, aimed at excluding secondary involvement from colorectal, prostatic, gynecologic, or urachal primaries through targeted imaging and clinical work-up [28, 31].
Definitive differentiation is based on characteristic microscopic findings:
Squamous cell carcinoma is defined by a pure squamous phenotype, including keratin pearl formation, intercellular bridges, and keratin debris, with no urothelial component [23, 26, 31].
Adenocarcinoma is characterized by true glandular differentiation, often with mucin production, and may exhibit enteric, mucinous, signet-ring, or mixed growth patterns [28, 31, 32].
Tumors containing any urothelial component are classified as urothelial carcinoma with squamous or glandular differentiation, rather than pure SCC or adenocarcinoma [28, 31].
The diagnosis of primary bladder adenocarcinoma is reserved for tumors with pure non-urothelial glandular morphology [28, 31].
Immunohistochemistry serves as a supportive tool, not a standalone diagnostic method:
In SCC, markers such as p40, p63, and CK5/6 confirm squamous differentiation but do not reliably distinguish pure SCC from urothelial carcinoma with squamous differentiation [23, 28, 31].
In adenocarcinoma, marker panels (e.g., CK7, CK20, CDX2, SATB2, β-catenin) assist in differentiating primary bladder adenocarcinoma from metastatic colorectal or Müllerian tumors [31-33].
Molecular and genomic profiling has demonstrated distinct mutational and immune-oncology signatures across bladder cancer subtypes; however, these approaches are not routinely required for basic histologic classification in current practice [32, 34].

Figure 2: Diagnostic differentiation between SCC and adenocarcinoma of the bladder.

Current treatments for bladder SCC mainly involve surgery, as systemic options are limited. For localized, non-metastatic disease, radical cystectomy with pelvic lymph node dissection is the standard, providing better survival than radiotherapy or chemotherapy. Radiotherapy, alone or combined, is reserved for unfit patients but yields worse outcomes. The carcinoma often resists chemotherapy, with no clear survival benefit from neoadjuvant or adjuvant therapies over surgery. In advanced cases, prognosis is poor, and treatment is usually palliative, often following urothelial carcinoma protocols. Emerging data suggest some activity of immune checkpoint inhibitors, especially before surgery, but evidence is limited. Early surgery and participation in clinical trials are strongly advised.
Radical cystectomy with pelvic lymph node dissection is widely regarded as the reference standard for the management of localized SCC and is associated with the most favorable survival outcomes compared with non-surgical approaches [36]. Evidence from large population-based analyses, systematic reviews, and institutional series consistently demonstrates that radical cystectomy, performed with or without perioperative chemotherapy, confers superior overall and cancer-specific survival compared with chemotherapy alone, radiotherapy alone, or combined chemoradiation in patients with nonmetastatic disease [37]. Long-term outcomes following surgery highlight the prognostic importance of tumor stage and nodal involvement, with durable disease-free survival reported in appropriately selected patients [37-39].
The benefit of peri-operative chemotherapy in node-negative localized SCC remains unproven, and available data suggest limited chemosensitivity in pure squamous histology, supporting a surgery-first approach in resectable disease [40]. Bladder-preserving strategies, including chemoradiation, have been explored in small retrospective series but are generally reserved for patients who are medically unfit for radical surgery or who decline cystectomy, given the weaker and less consistent supporting evidence [41]. Partial cystectomy is considered only in highly selected cases with favorable anatomy and requires stringent long-term surveillance [41]. Overall, current evidence supports early radical cystectomy with lymphadenectomy as the guideline-concordant primary treatment for non-metastatic bladder SCC, with non-surgical alternatives limited to selected clinical scenarios [42].
For patients with localized or locally advanced SCC of the bladder, surgery, particularly radical cystectomy with pelvic lymph node dissection, represents the cornerstone of curative treatment, while other therapeutic modalities generally serve adjunctive or alternative roles [37, 43]. Large population-based analyses from SEER and the National Cancer Database consistently demonstrate that radical surgery is associated with superior overall and cancer-specific survival compared with radiotherapy alone, observation, or other non-surgical approaches in non-metastatic disease [44]. Survival benefits with surgery are observed in both localized and regional (locally advanced) stages, although the magnitude of benefit decreases in the presence of distant metastases, where surgery is largely palliative [44, 45].
Bladder-preserving approaches, including partial cystectomy or TURBT-based strategies, have been reported only in highly selected cases and are supported primarily by limited retrospective data [46]. Long-term outcomes from population studies indicate that these approaches generally provide inferior cancer-specific survival compared with radical cystectomy and require intensive lifelong surveillance [36].
The addition of perioperative systemic therapy has not shown a clear survival benefit in node-negative localized SCC and is therefore not routinely recommended; however, in patients with very high-risk features—such as node-positive, T4b, or metastatic disease—combined surgical and systemic approaches may reduce cancer-specific mortality [37, 40, 47]. In bilharzial-associated SCC, selected series suggest a potential role for perioperative radiotherapy to improve local control, though the quality of evidence remains limited. Overall, early radical cystectomy remains the most effective curative strategy for localized and locally advanced bladder SCC, with non-surgical options reserved for carefully selected or surgically unfit patients [26, 36].
For bladder SCC, radical cystectomy should almost always include bilateral PLND, as omission clearly worsens survival. Pelvic lymph node dissection (PLND) clearly improves survival in bladder SCC when performed with cystectomy, but current data support a “standard” rather than an extended template. In a SEER analysis of variant histology, PLND versus no PLND significantly improved overall survival in SCC (HR ~0.65), with median OS nearly doubled (≈71 vs 37 months) [48]. A large NCDB study of variant bladder cancers confirmed that lymph node dissection is independently associated with better OS in the squamous subtype (HR 0.50), one of the strongest benefits among variants [49]. Despite this, SCC has high pNx rates (~68%), indicating PLND is underused in this histology [49].
Evidence on extent comes from mixed histology (mostly urothelial) MIBC, but no data suggest SCC behaves differently:
Two RCTs and multiple meta-analyses show no improvement in overall or disease-free survival with extended PLND to the aortic bifurcation compared with standard PLND up to the common iliac bifurcation [50, 51].
Extended PLND increases major complications, lymphoceles, and 90-day mortality [50].
SEER analysis in variant histology found greater PLND extent did not reduce cancer-specific mortality in VHBC (including SCC), although it improved staging yield [52].
For variant histologies, PLND remains prognostic even when chemo or neoadjuvant therapy is used, but added survival from “more extensive” dissection diminishes after neoadjuvant chemotherapy [53]. Immunotherapy outcomes (pembrolizumab) do not appear improved by more extensive node harvest, and >14 nodes after NAC may even correlate with worse progression-free survival in mixed histology cohorts [54].
Definitive radiotherapy is feasible for non‑surgical bladder SCC, but expected cure rates are low; concurrent chemoradiation is preferable to RT alone when possible. Non-bilharzial SCC of the bladder is aggressive and usually treated with radical cystectomy. For patients who cannot undergo surgery, evidence for definitive radiotherapy (RT) is limited and mostly retrospective, but offers a potentially curative alternative to best supportive care.
In the largest NCDB analysis of localized cT2–3N0 pure SCC, RT alone and chemoradiation (CRT) had substantially worse overall survival (OS) than cystectomy (HR for death vs RC: RT 4.78; CRT 1.61) [37]. A SEER study of 5653 SCC cases found median survival 12 months with RT, 9 months with chemotherapy, vs markedly better with surgery; adding RT to surgery did not improve OS, likely reflecting selection bias [44]. Historical RT alone series in SCC report ~16% 3 year disease free survival with 60–65 Gy, indicating poor local control [55].
NCDB CRT analysis (79 SCC, cT2–T4N0M0) using concurrent Chemoradiation (CRT) found median OS ~15 months for SCC vs ~29–30 months for urothelial carcinoma; SCC histology independently predicted worse OS [55]. Case-based and small series suggest some long-term survivors using 5 FU/mitomycin or cisplatin-based CRT, extrapolating from anal SCC and urothelial CRT trials [36, 55].
Guidelines for MIBC support definitive RT or CRT in cystectomy-unfit patients, acknowledging lower-level evidence, and this is often extrapolated to SCC [56]. Narrative and systematic reviews emphasize that RT with concomitant chemotherapy is a reasonable alternative for unresectable tumors or in patients seeking bladder preservation, but data specific to SCC are sparse and of low level [36]. In broader MIBC cohorts unfit for surgery or cisplatin, RT (often without chemo) substantially improves OS vs supportive care, supporting its use as active treatment in frail patients [57].
In bladder SCC patients not suitable for cystectomy, a bladder-preserving approach with TURBT followed by chemoradiotherapy is preferred. Curative radiotherapy often involves doses ≥60 Gy with radiosensitizers like 5-fluorouracil, mitomycin C, cisplatin, or weekly gemcitabine. While this may control disease in some, cure rates are lower than with cystectomy [36]. For those unable to have chemotherapy, definitive radiotherapy alone at radical doses may improve survival versus no treatment, but outcomes are limited and palliative [44]. Due to high local recurrence risk, close surveillance with cystoscopy and imaging is vital, and salvage cystectomy should be considered if the patient’s condition and fitness improve [36, 55].
Adjuvant radiotherapy (ART) after radical cystectomy is being revisited because loco-regional recurrence rates for ≥pT3 disease remain high, and salvage is difficult. Modern intensity modulated techniques have reduced historical toxicity, enabling safer postoperative treatment. High risk features associated with loco regional failure include pT3–4, pN+, positive margins, and limited lymph node dissection (<10 nodes) [58, 59]. Phase II–III and retrospective studies consistently show substantial reductions in pelvic relapse with ART or chemoradiotherapy compared with observation or chemotherapy alone, especially in ≥pT3/4 or margin-positive disease [60, 61].
With IMRT/VMAT, severe GI toxicity is markedly lower than in historical 2D RT: grade ≥3 GI events ~3–6%, though grade 2 GI symptoms are common. Phase II trials show acceptable urinary toxicity even in neobladder patients [62].
Reviews and guidelines suggest that ART is reasonable for selected high-risk MIBC, especially with positive margins, pT3–4, pN+, or suboptimal node dissection, and where access to salvage or immunotherapy is limited [63]. Large databases show no consistent OS benefit overall, but a possible benefit in marginally positive patients [64, 65]. International consensus CTV guidelines now recommend including the cystectomy bed and common iliac nodes for high-risk cases [66].
Most work on neoadjuvant radiotherapy (RT) in bladder cancer is in mixed or predominantly urothelial histology. For pure bladder squamous cell carcinoma (SCC), evidence is largely indirect, coming from older series or reviews that pool histologies. A systematic review of bladder SCC notes that preoperative RT + radical cystectomy (RC) appears to improve survival compared with RC alone, but the data are from small, older series and are insufficient for firm recommendations. The same review highlights SCC’s high local recurrence risk and suggests neoadjuvant or adjuvant RT (± systemic therapy) may reduce recurrence, but emphasizes the lack of randomized SCC-only trials [36].
Contemporary reviews of perioperative RT in muscle-invasive bladder cancer argue that preoperative RT is attractive in very high-risk, locally advanced disease to sterilize microscopic extension and improve pathologic response, but call the evidence “preliminary” and non-definitive [67]. Current neoadjuvant RT trials (often with immunotherapy such as nivolumab or durvalumab) are in urothelial carcinoma, not SCC; they explore feasibility and pathological complete response but do not yet guide SCC practice [68].
For operable, locally advanced SCC, RC remains standard; neoadjuvant RT can be considered only in selected very high-risk cases, extrapolating from mixed histology data and acknowledging the lack of SCC-specific proof [36, 38]. For patients unfit for RC, management usually shifts to definitive chemoradiation, not “neoadjuvant” RT, again extrapolated from urothelial MIBC paradigms rather than SCC trials [69].
For bladder squamous cell carcinoma, radical cystectomy remains standard; chemoradiation is mainly a bladder‑sparing or non‑surgical option and appears less effective for survival but can offer local control when surgery is not feasible.
A large NCDB study of 828 patients with localized muscle-invasive pure SCC (cT2–3N0M0) compared RC ± perioperative chemotherapy, chemotherapy alone, radiation alone, and definitive chemoradiation. RC (with or without chemo) had the best survival; chemoradiation carried a higher death risk (HR 1.61 vs RC alone) but was better than radiation alone or chemotherapy alone [37]. A systematic review of non-bilharzial SCC concluded RC is the gold standard; radiotherapy alone gives poor outcomes, while multimodality approaches (pre op RT + RC, or RC + systemic therapy) may help, but evidence is low-level and heterogeneous [36].
Variant histology (including SCC) shows worse survival after bladder preserving RT than pure urothelial carcinoma, highlighting more aggressive biology and the need for intensification or surgery when possible [55].
Indications of chemoradiation in Bladder SCC:
Regimens generally mirror urothelial protocols: conventional or hypofractionated pelvic RT (≈55–64 Gy) with concurrent cisplatin, gemcitabine, or 5 FU/mitomycin C as radiosensitizer [56]. Given chemotherapy resistance concerns in SCC, systemic control may still be suboptimal, and close surveillance or consideration of combined RC, where feasible, is important [36, 55].
All Rights Reserved to Indigoarabia.com
Stay Connected for latest Genitourinary news
connect with us on LinkedIn for professional updates and collaborations.
All Rights Reserved to Indigoarabia.com